
Home » DxTerity Earns CE Mark for Finger Stick Blood Collection Tube
DxTerity Earns CE Mark for Finger Stick Blood Collection Tube
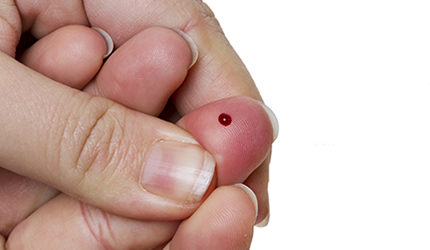
DxTerity received CE Mark certification for its DxCollect MicroCollection tube, a device used to collect and store finger stick blood samples that are used in genomic testing.
The tube is capable of transporting up to 100 microliters of blood taken with a finger prick for DNA and RNA-based genomic testing.
The device can stabilize samples at room temperature and can be shipped in ambient conditions using standard mail, the company said.
Upcoming Events
-
11Jul
-
18Jul
-
21Oct

