
Home » OPTI Medical’s COVID-19 Test Kit Gains CE Mark
OPTI Medical’s COVID-19 Test Kit Gains CE Mark
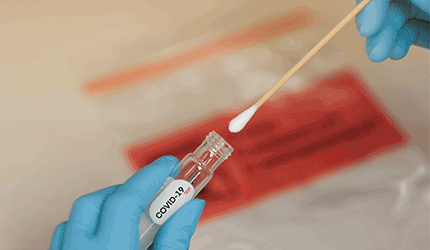
OPTI Medical has earned the CE mark for its OPTI SARS-CoV-2 RT-PCR laboratory test kit for detecting the coronavirus.
The polymerase chain reaction (PCR) test detects the virus from nasopharyngeal swabs, oropharyngeal swabs, sputum samples and bronchoalveolar lavage, and it returns results in approximately two to 3.5 hours. The test runs on commonly available PCR instruments.
The diagnostic has already received an EUA from the FDA.
Upcoming Events
-
11Jul
-
18Jul
-
21Oct

